Washington, DC (UroToday.com) Dr. Arnulf Stenzl provided the EAU guest lecture today at the 20th Annual Meeting of the Society of Urologic Oncology, discussing his innovative work on improving urologic oncology surgery with intraoperative real-time technology. Dr. Stenzl has more than 600 publications and two international patents. He is the former chair of the EAU Scientific Congress and is on the executive board of the Comprehensive Cancer Center of Southwestern Germany. His research and clinical interests include bladder and prostate cancer, as well as reconstructive surgery.
Dr. Stenzl notes that currently, our gold standard for intraoperative diagnostics is the frozen section. However, he highlights that this is troublesome considering that the specimen leaves the OR (costing OR time), the flow of surgery is interrupted, and there are issues with specimen selection vs tissue heterogeneity, as well as the micro-extent of the tumor. The aim would be for the specimen to not leave the OR, have a real-time assessment, and sample multiple specimens in an effort to decrease heterogeneity of findings. Frozen sections are only positive 5% of the time, however, there is a 25% false negative rate. Standardized frozen sections during a radical prostatectomy lower the positive margin rate from 22% to 15% but have no bearing on biochemical recurrence rates, and the effect on functional results is unknown.
We have recently seen the effect of PET guided salvage lymphadenectomy, with PSMA PET-CT scans identifying lymph node metastasis with 77.9% sensitivity, leading to PET-guided salvage lymphadenectomies resulting in PSA < 0.2 ng/dL in 56% of cases. But, Dr. Stenzl notes, that we are relying on PSMA PET-CT for cognitive localization. What we need to get to is PSMA radio-guided surgery:
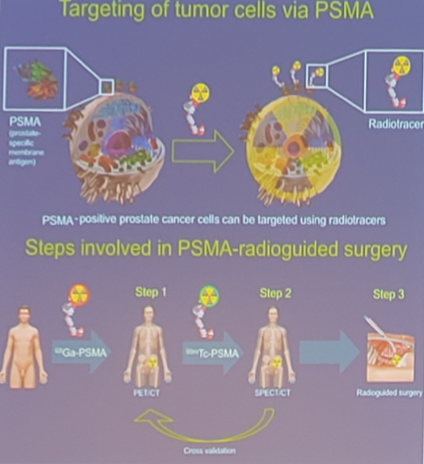
We do already have open radio-guided surgery, particularly as it has been used in axillary dissections for breast cancer. This technology has also recently been adapted to the identification of sentinel nodes in robotic surgery with a drop-in gamma probe through one of the working ports. Furthermore, in mouse models, PSMA-labeled fluorescence improves the detection of prostate cancer.
Another technological advance Dr. Stenzl highlighted is tissue differentiation by optical emission spectroscopy. The aim of this is to apply the technology for assessing tissue recognition for renal cell carcinoma. This provides real-time analysis of electrosurgical sparks, providing information on tissue type in the operative setting. From preliminary data, Dr. Stenzl notes that this technology is able to detect renal cell carcinoma histology with a sensitivity of 96%, a specificity of 93%, positive predictive value of 94%, and negative predictive value of 96%.
Raman spectroscopy is able to differentiate benign from malignant tissue down to the cellular level by assessing the level of DNA methylation; sensors have also been adapted for use during robotic surgery. Discrimination of tissue has also been reported via infrared-spectroscopy, taking advantage of changes in cell metabolism altering light absorption in the infrared spectrum.
Dr. Stenzl notes that discrimination of deeper tissue layers has been described using elastography and model-based optical sensors, using optical methods to measure the changing stiffness of the extracellular matrix. This technology takes advantage of optical coherence tomography, holography, and speckle to compare measurements using a finite element model.
Ultimately, all of these technological advances rely on intraoperative navigation, correlating measured sensor data to the anatomical location and identifying the location of interest for the surgeon. Dr. Stenzl concluded noting that intraoperative uro-oncological real-time diagnosis provides precision tumor ablation, while at the same time limiting collateral tissue damage. Although a tumor frozen section is currently the intraoperative gold standard, optical emission spectroscopy, Raman spectroscopy, infrared spectroscopy, single-cell elastography, model-based optical sensors, electric impedance spectroscopy, and tissue elastography may all have the ability to differentiate tumor from normal tissue in real-time. These advances may allow intraoperative localization of measured data and guide surgical approaches.
Presented by: Arnulf Stenzl, MD, Professor and Chairman, Department of Urology, University Hospital Tubingen, Tubingen, Germany
Written by: Zachary Klaassen, MD, MSc – Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md at the 20th Annual Meeting of the Society of Urologic Oncology (SUO), December 4 - 6, 2019, Washington, DC

